In recent years, many people have come across or at least heard of such a phenomenon as acid rain.
Such an atmospheric phenomenon occurs due to pollution of the atmosphere and water by various man-made pollution. The first recorded acid rain fell in 1872.
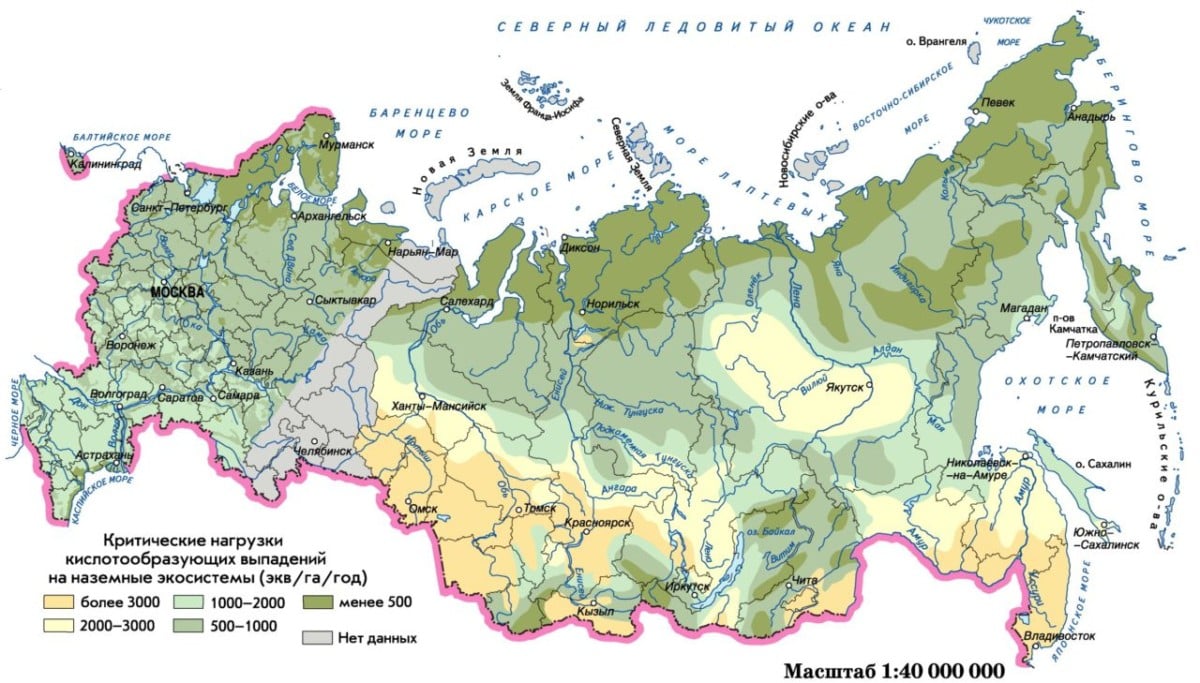
Acid-prone areas
In the last century, especially in its last decades, this phenomenon was no longer something very unusual. Where does acid rain form? Such rains (and this concept also includes acid hail, and the same snow) fall with relative regularity in Europe, and in Russia, and in the USA.
Ecologists have developed a map, which shows in detail each area exposed to such dangerous phenomena.
On the territory of most constituent entities of the Russian Federation, the precipitation of sulfur and nitrate nitrogen from own sources does not exceed 25 percent of their total precipitation. The contribution of own sulfur sources exceeds this threshold in the Murmansk (70%), Sverdlovsk (64%), Chelyabinsk (50%), Tula and Ryazan (40%) regions and in the Krasnoyarsk Territory (43%).

In the European territory of Russia, only 34 percent of sulfur deposits are of Russian origin. Of the remainder, 39 percent comes from European countries and 27 percent from other sources. At the same time, neighboring countries make the greatest contribution to transboundary acidification of the natural environment: Ukraine, Poland, Germany, Belarus and Estonia.
What is acid rain
The normal value is 5.6. If gases interact with water, then the indicator drops to 1–4. This causes the death of microorganisms. Cells of more complex organisms also die.
It is recognized that acid rain is a global environmental problem. They go more and more often. This cannot but cause concern for both the scientific community and the public. The fallout of toxic precipitation adversely affects the flora and fauna of all lands.
People also suffer from this. Sometimes it’s enough just to be exposed to acid rain and get a lung or heart disease.
As already mentioned, acids can be present not only in rain, but also in snow, in hail. Moreover, they can even be in the fog. At the same time, the higher the concentration of any impurities in the moisture, the more harm it causes.
Most often, laboratory tests are required to detect acid in atmospheric moisture. But sometimes this is evidenced by the burning sensation that appears in a person when breathing and even on the skin. In many cases, such precipitation has a sour smell. A change in the color of the water is also a sign of pollution. If in such conditions to be in open space, then everything can end even with acid burns.
Acid rain is dilute acid. Because of this, the air and soil (primarily the fertile layer) are saturated with toxins. Because of the acid, animals and plants die, minerals are destroyed, food products become simply poisonous.
The revival of the affected area has been going on for a long time. Acid, penetrating into the soil, gradually sinks into the depths, contaminating groundwater.
Causes of acid precipitation
It is considered the norm when weak carbonic acid is present in precipitation. This is recognized as a natural state. The fact is that there is always carbon dioxide in the air, and this acid is formed when it interacts with moisture. Abnormal precipitation is formed in a similar way. But at the same time, moisture interacts with other volatile compounds. As a result, the clouds are saturated with sulfuric or nitric acid. At the same time, the more the air is polluted with sulfur compounds or simply sulfur, or nitrogen compounds, the higher the concentration of acid in the precipitation.
Among the reasons for abnormal precipitation are the following:
- Volcanic eruptions release ash and various gases into the atmosphere. These are sulfur oxides, and sulfates, and hydrogen sulfide substances.
- During the decomposition of organic residues, both animals and plants, sulfur and nitrogen compounds enter the atmosphere.
- The lightning discharge also provides natural atmospheric pollution. It is recognized that annually due to lightning, the volume of acids formed reaches eight million tons.

Currently, air pollution is mainly due not to natural factors, but due to human activities. The scientific community recognizes that the anthropogenic impact in this regard is now between 89 and 92%. Air pollution is caused by:
- emissions from enterprises operating in mechanical engineering and metallurgy, thermal power plants;
- when burning coal, oil products, gas, firewood;
- due to the release of methane, which occurs in the fields where cereals are grown;
- due to the use of fertilizers containing nitrogen, as well as the treatment of crops with pesticides;
- in mining, especially in open pit mining;
- freons released into the atmosphere from faulty refrigerators;
- when using aerosols containing hydrogen chloride.
How acid rain is formed
The mechanism of acid precipitation formation can be simply explained by chemical formulas. A significant role in the formation of acid precipitation is played by emissions of harmful substances that occur during the operation of mining and processing enterprises. These emissions contain up to 65% of sulfur and its compounds. They dissolve in moisture.
Sulfur Rain
When sulfur dioxide enters the atmosphere, a reaction called photochemical oxidation occurs with the formation of sulfuric anhydride. The next reaction is its interaction with water vapor, resulting in the formation of sulfurous acid. But the process doesn’t stop there. The interaction of this compound with water continues, and this acid is converted into sulfuric acid.
Nitrogen Rain
Half of acid rain is nitrogen. In this case, the formation of rainwater occurs due to nitrogen-containing compounds. Dissolving in steam, they become nitrous or nitric acid. When such rain falls on the ground, the chemical reaction continues in the soil. This results in the formation of nitrates and nitrites.
Salt Rain
It happens that under the influence of compounds containing chlorine, salt rains are formed. This gas is in the air at the same time as methane. This happens during emissions from a number of industrial enterprises and during the burning of garbage.
In addition to these substances, compounds that are highly toxic also enter the atmosphere. Industrial enterprises are also the culprits. The evaporation that occurs during the decomposition of household waste also contributes its share.
Consequences of acid rain
Acid rain can harm entire regions. The time of complete decomposition of toxic compounds that have entered the soil or water reaches 15 years. What harm does acid rain do to plants? Once in the soil, these substances destroy everything that plants need. This releases metal salts such as lead, aluminum and mercury. On such soil, plants simply cannot exist. If a highly concentrated acid is present in the rain, mature plants and seeds die. At the same time, some of the toxic compounds poison the air.

Global Issues
Under the influence of acid rain, such global environmental problems appear:
- The structure of water changes. Its rigidity increases, water is saturated with toxins and acids. Acid rain is the most dangerous for flora and fauna. as a result, animals and plants die. It happens that after such rains, the content of heavy metals in water is 8 times higher than the maximum permissible norms.
- Due to the fact that the leaves and root system of plants are damaged, the plants die. The consequence is the death of herbivores, which forces predatory animals to move to other territories. That is, food chains are broken.
- Contaminated topsoil due to winds continues to pollute the air. The wind carries poisonous dust over considerable distances, poisoning nature there as well.
Influence on a person
Acid rain also has a negative effect on humans. Interacting with a number of building materials, they release polycarbonates from them. Destruction reaches such a level that these substances, as well as air moisture, begin to oxidize the metals present in the structures. Because of this, buildings quickly collapse. Due to acid rain, livestock is dying, crops and inhabitants of reservoirs in fisheries are suffering.
Under the influence of acid rain, people develop diseases such as: bronchitis, pneumonia, tracheitis, bronchial asthma. In some cases, people even get chemical burns.
Unfortunately, this is not the end of the matter. Meat products, fish, products made from plants, are saturated with mercury, lead, sulfur, arsenic, selenium are harmful to human health. Both acids and salts are stored in products of animal origin, due to which food poisoning occurs. In this case, the liver, pancreas, kidneys, circulatory and nervous systems suffer.
Economic implications
Acid rain also causes significant damage to the economy. Significant costs are required to compensate for the damage caused to agriculture, forests, and water bodies. Since crop areas also suffer, crop losses reach large values.
The consequence may be famine, especially when it comes to underdeveloped regions.
Since, due to chemical burns, diseases caused by harmful compounds that have entered the body, there is a need to provide medical care in significant volumes, the costs of medicines and the work of medical staff increase. The cost of compensation for the consequences of such a program alone reaches tens of billions of dollars.
Measures against acid precipitation
The development of measures aimed at preventing harm from acid rain is being carried out in the United States, in Russia, and in China. The reason is that there are a large number of coal mining enterprises in these countries. Many and metallurgical enterprises.

Fighting environmental problems on a local scale is impossible. We need an integrated approach to reduce harmful emissions, acid-forming substances into the atmosphere, water and soil, which is possible only with interstate cooperation. The use of alternative energy sources without the use of coal, oil and gas also leads to a reduction in acid precipitation. The use of low-sulfur coal or its purification from sulfur will also help. Such work is underway.
Designers develop high-performance air filters and cleaning systems. However, such problems cannot be solved in a short time. The negative effects of atmospheric pollution with subsequent acid rain continue to increase every year, many large industrial enterprises are responsible for the formation of acid rain.
Without an appropriate analysis, it is impossible to determine whether acid is present in rain, snow, fog. Therefore, if a warning about the threat of acid precipitation is received, the doors and windows of residential buildings and industrial premises should be especially carefully closed. In such cases, volatile compounds are always present, which can have a harmful effect even when a person is not exposed to rain or snow.